Project Updates
CoreLink’s Siber Ti Sacroiliac Joint Fusion System, US

Credit: CoreLink, LLC.
Project type: Sacroiliac joint fusion system
Developer: CoreLink
Applications: For sacroiliac joint fusion by surgical procedures
Status: Approved in the US
The Siber Ti Sacroiliac (SI) Joint Fusion System is a family of fully porous, nano-surfaced, 3D-printed implants developed by CoreLink, to support surgeons in SI joint fusion procedures. It received 510(k) clearance from the US FDA in March 2023.
Stryker’s Q Guidance System, US
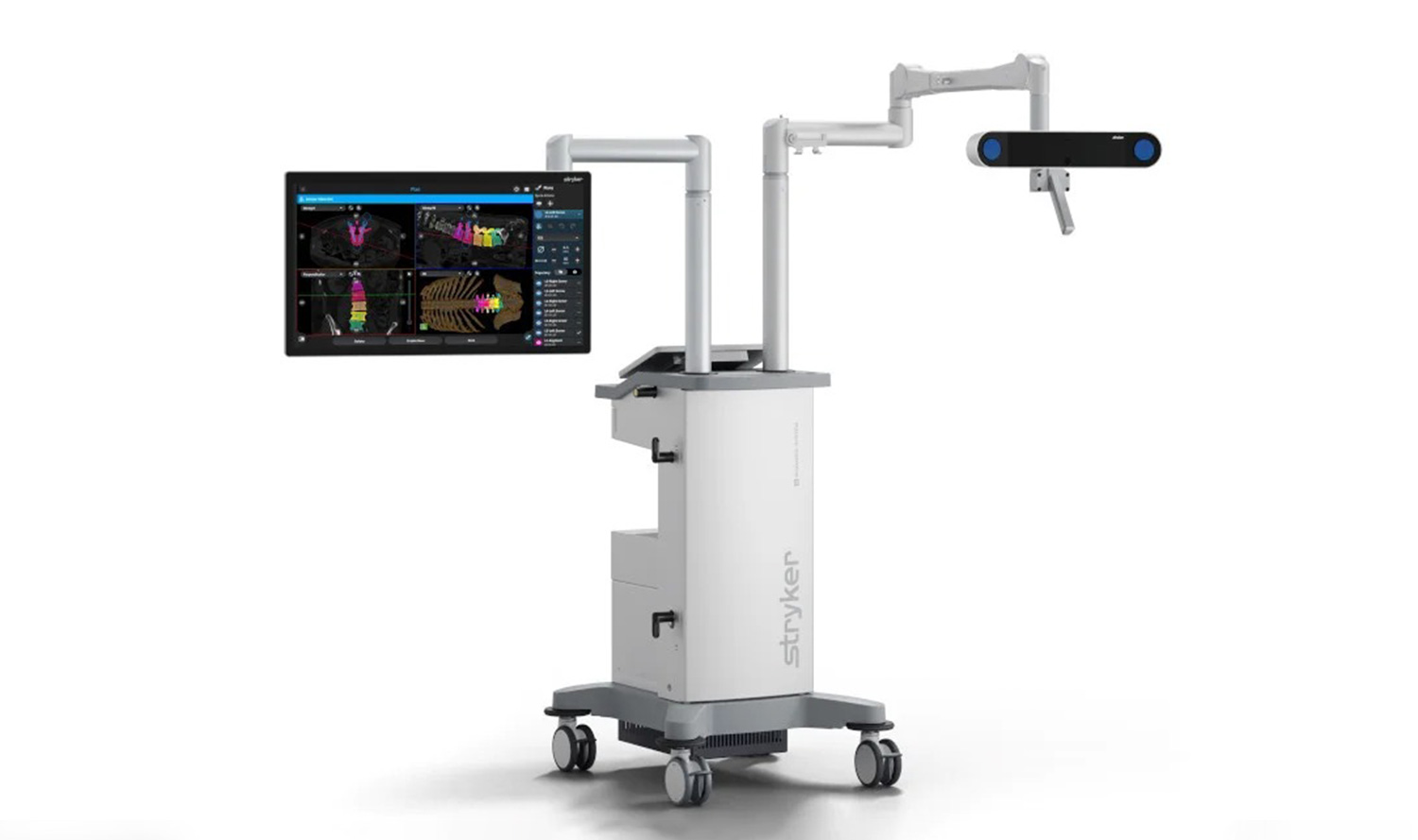
Credit: Stryker
Project type: Image-based planning and intraoperative guidance system
Developer: Stryker
Applications: To support cranial and spinal navigation and surgeries
Status: Approved in the US
The Q Guidance System is an image-based planning and intra-operative guidance system developed by Stryker, to support spine and cranial surgery procedures. It received 510(k) clearance from the US FDA in June 2022.
LivaNova’s Essenz Heart-Lung Machine, UK

Credit: Business Wire
Project Type: Perfusion system
Developer: LivaNova
Applications: Cardiopulmonary bypass management
Status: Approved in the US and UK
The Essenz Heart-Lung Machine was developed by UK company LivaNova to support open-heart surgery procedures. The next-generation Essenz system with embedded Essenz Patient Monitor forms the state-of-the-art Essenz Perfusion System for safe cardiopulmonary bypass management.