DEALS ANALYSIS
Deals activity: YoY growth for Asia-Pacific; licensing agreement deals are up
Powered by
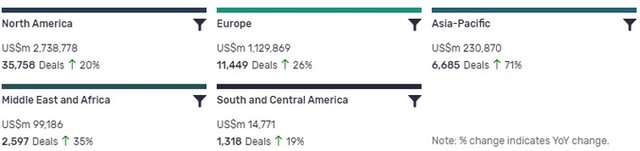
Deals activity by geography
Medical industry deals, as captured by GlobalData’s Medical Intelligence Centre, are up year-on-year (YoY) across all regions.
North America is leading in terms of deal value, but recorded the second lowest YoY growth in deals volume at 20%. Asia-Pacific, ranking third in terms of deal value, has seen the biggest YoY change, with deal volumes increasing by 71%.
The volume of deals recorded by GlobalData also increased YoY in Europe (26%) and Middle East and Africa (35%), but declined in South and Central America (19%)
Deals activity by type
| Deal type | Total deals value ($m) | Total deal count | YoY change (volume) |
| Venture Financing | 140,584 | 16,621 | 58 |
| Partnership | 14,904 | 10,687 | 21 |
| Equity Offering | 354,484 | 8,040 | 142 |
| Acquisition | 1,984,369 | 7,172 | -45 |
| Debt Offering | 1,104,258 | 2,394 | 5 |
| Asset Transaction | 99,677 | 1,793 | -47 |
| Private Equity | 265,148 | 1,693 | 90 |
| Grant | 1,019 | 1,902 | 128 |
| Licensing Agreement | 6,606 | 676 | 721 |
| Merger | 147,346 | 295 | 22026 |
A breakdown of deals by type and volume shows a 22026% growth in mergers YoY, while acquisitions are down 45%, partnerships are up 21% and asset transactions are down 47%. Financing deals have increased across some types, with venture financing up 58% YoY, equity offerings up 142% and debt offerings up 5%.
Private equity has grown by 90% in number of deals YoY, while the number of grants recorded is up 128%.
Deals activity by therapy area
A notable development apparent in GlobalData’s analysis of medical industry deals by therapy area is the increase of deals in the field of healthcare IT. After remaining relatively steady, the number of recorded deals healthcare IT increased significantly in 2020. This is likely due to demand caused by the ongoing Covid-19 pandemic.
Note: All numbers as of 03 January 2021. Deals captured by GlobalData cover M&As, strategic alliances, various types of financing and contract service agreements.
For more insight and data, visit GlobalData's Medical Intelligence Centre
Latest deals in brief
Honeywell to acquire Sparta Systems for $1.3bn
Industry-specific solutions provider Honeywell has agreed to acquire Sparta Systems and its med tech and pharma quality management software for $1.3bn from New Mountain Capital. With this acquisition, Sparta’s AI-enabled software as a service quality management software offering will be merged with an enterprise performance solution, Honeywell Forge and Experion Process Knowledge System to boost value for life sciences customers in the US, Europe and Asia.
Philips, BioIntelliSense to receive US funding for early detection of Covid-19 symptoms
Royal Philips and BioIntelliSense are set to receive about $2.8m funding from the US Department of Defense to validate BioIntelliSense’s FDA-cleared BioSticker device for the early detection of symptoms related to Covid-19. The companies will get the funding through a Medical Technology Enterprise Consortium award.
PRA, PWNHealth and Fulgent Genetics partner for at-home Covid-19 test
Contract research organisation PRA Health Sciences has enhanced its Covid-19 Monitoring Program by collaborating with PWNHealth and Fulgent Genetics to provide at-home Covid-19 testing capabilities. The capabilities will add to PRA’s current Covid-19 clinical monitoring, symptom reporting, and management services accessible on the patient-facing Health Harmony mobile app.
Sherlock Biosciences gets grant to develop Covid-19 diagnostic platform
Engineering biology company Sherlock Biosciences has received a grant worth $5m from the Bill & Melinda Gates Foundation to advance the development of its instrument-free, synthetic biology-based molecular diagnostics platform. The Internal Splint-Pairing Expression Cassette Translation Reaction platform uses synthetic biology to aid in creating instrument-free diagnostic tests that can be carried out at home and room temperature.
CSIR partners with Apollo to launch dry swab Covid-19 test in India
Molecular Biology (CSIR-CCMB) in India has collaborated with Apollo Hospitals to manufacture and commercialise a novel dry swab RT-PCR test. Known as Direct Amplification rapid Reverse Transcription – Polymerase Chain Reaction for SARS-CoV-2 detection, the test is rapid, safe, and cost-effective.
Roche partners with Moderna to include Covid-19 test in vaccine trials
Roche has entered a partnership with Moderna to use the Elecsys Anti-SARS-CoV-2 S antibody test in research trials of the latter’s Covid-19 vaccine, mRNA-1273. The collaboration will support the quantitative measurement of SARS-CoV-2 antibodies. It will also help to establish a connection between protection induced by the vaccine and levels of anti-receptor binding domain antibodies.